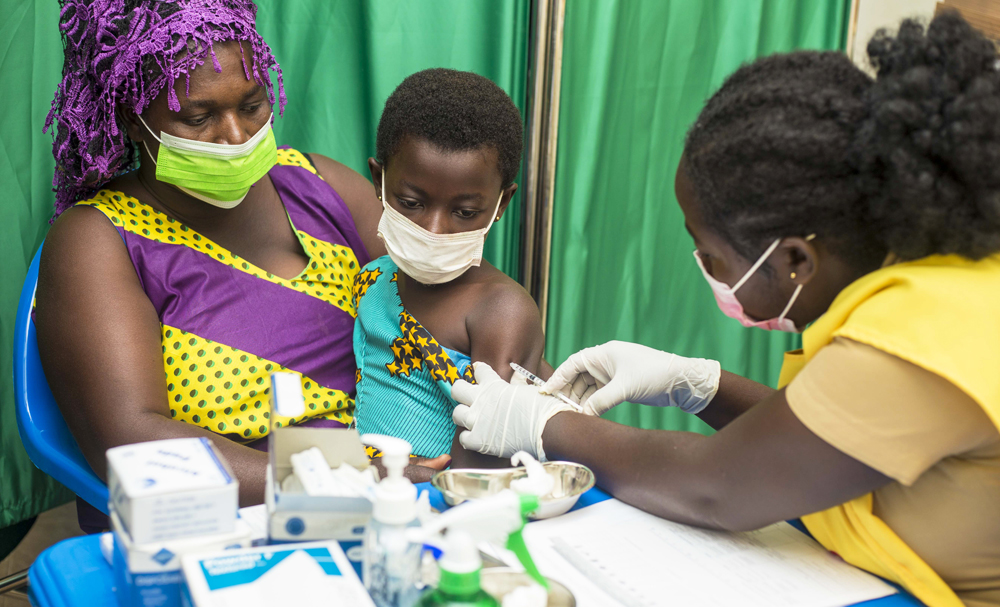
The Kwame Nkrumah University of Science and Technology in collaboration with the International Vaccine Institute (IVI) has commenced a vaccine trial for Typhoid fever at Agogo in the Asante Akim North Municipal District of the Ashanti Region on Monday, 23rd August 2021. The vaccine will be administered to 23,000 children aged between nine months and fifteen years to gain the necessary immunity to fight typhoid fever.
The Typhoid Conjugate Vaccine Effectiveness in Ghana (TyVEGHA) trial aims to introduce the typhoid Vi polysaccharide conjugate vaccine using tetanus toxoid as the carrier protein. This vaccine has been prequalified by WHO in 2018 to offer overall protection from Typhoid infection for people living in endemic areas such as the Asante Akim North District of the Ashanti Region and subsequently for further deployment to other parts of Ghana and Africa, after immunogenicity and safety data are analysed.
The trial is being conducted by a team of scientists led by Professor Ellis Owusu-Dabo from the School of Public Health, Kwame Nkrumah University of Science and Technology and in Korea by Dr. Florian Marks, Deputy Director General, Epidemiology, Public Health and Impact and Clinical Development at IVI, South Korea with high-level strategic input and support from other collaborating institutions such as University of Cambridge, Fondation Mérieux, Bill and Melinda Gates Foundation, and other partners from Democratic Republic of Congo, Burkina Faso and Madagascar, with Agogo Presbyterian Hospital as the trial site for Ghana.
The programme is being funded by European Developing Countries Clinical Trials Partnership (EDCTP) and the Bill and Melinda Gates Foundation. The state-of-the-art trial centre at Agogo Presbyterian Hospital has been built with support from KNUST, Agogo Presbyterian Hospital, and the funders.
According to the Principal Investigative Officer of the project, Professor Yaw Adu Sarkodie, the vaccine trial will help in generating additional data to aid decision-making on its local adoption for the management of typhoid and other fevers in the sub-region. It will also help develop solutions to counter poverty-related diseases which have a huge economic impact on populations in West Africa.
He noted that the trial would go on for a period of three years with keen observation, after which the Ghana Health Service (GHS), together with the Food and Drugs Authority (FDA), will ascertain the potency of the vaccine and recommend it for use in the country.
Professor Kwame Ohene Buabeng, Head of Department of Pharmacy Practice KNUST, assured the public that the vaccine had undergone several trials in other countries and is safe for human use.
Typhoid fever is a highly contagious disease that is mostly spread through contamination of food and water. Early detection of the disease can be treated with a course of antibiotics. However, if it is left untreated, the disease can become severe and even fatal.
Typhoid fever is an infectious disease that is caused by a species of bacteria called Salmonella typhi that affects low-and middle-income countries.